An Introduction to Cavernous Malformation
by Joseph M. Zabramski, MD (Professor Neurological Surgery, Barrow Neurological Institute, Phoenix, AZ).
The goal of this article is to provide a basic introduction to cerebral cavernous malformations. This will include a description of these lesions, their frequency in the adult population, the risks they pose to those effected, and the presently available treatment options.
Before we start, it is important to recognize that these lesions are known by multiple names, including cavernomas, cavernous angiomas, cavernous hemangiomas, cavernous malformations, and intracranial vascular malformations. The terms cavernous malformations and cerebral cavernous malformations (abbreviated CCM) are the most common and will be used here.
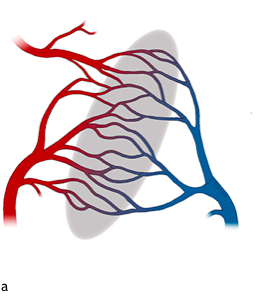
What are CCMs? Cavernous malformations are vascular lesions found throughout the brain and spinal cord. They typically measure from 1 to 5 cm in size, but lesions as small as 1 to 2mm are not uncommon. They are composed entirely of a tangle of small, abnormal blood vessels. Structurally the blood vessels in these lesions resemble the tiny vessels called capillaries which carry blood from the arteries to veins providing oxygen and glucose to our tissues and carrying away waste products (Figure 1a).
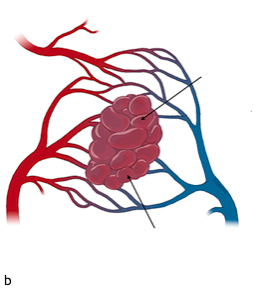
Unlike normal capillaries, the vessels that make up cavernous malformations have weakened walls that become dilated (cavernous, Figure 1b). Blood within these dilated vessel segments stagnates and forms thrombus (clot). The subsequent breakdown of the clotted blood releases hemoglobin and other chemicals from the red blood cells that escape through the abnormal vessel walls into the surrounding tissue, resulting in the classic appearance of these lesions on magnetic resonance imaging (more on that below). Much less frequently the walls of the vessels that make up the cavernous malformation will rupture open allowing blood to escape directly into the surrounding brain creating a localized hematoma (a blood clot).
Who is affected? CCM lesions a much more common than generally appreciated. Approximately 0.5% of the adult population harbors CCM lesions. Looked at another way, about 1 in every 200 adults is affected with this disease. Fortunately, most of these cases remain silent and never cause any symptoms. So, while more than 12 million people in the United States are affected by cavernous malformations, only about 200,000 have suffered a symptomatic hemorrhage.
Cavernous malformations occur in two forms: 1.) A familial, hereditary form that makes up 20% to 30% of symptomatic cases and presents with multiple lesions with a strong family history of seizures and stroke, and 2.) A more common sporadic form, that typically manifests as a solitary lesion with no family history. While patients with the familial form make up only about 5% of patients with cavernous malformation, but because they have multiple lesions, they are more likely to present with symptoms.
Genetic inheritance: The familiar form of CCM is an inherited disease, being passed down from one generation to another as a genetic mutation. For patients with the familial form of CCM the mutation that causes the disease is inherited in an autosomal dominant fashion; that means that there is a 50:50 chance that children of an affected parent will also have the disease.
Three different genetic mutations have been identified that cause the familial form of cavernous malformations: CCM1, CCM2, and CCM3. It is thought that the products of these three genes form a complex that is necessary for the development of normal blood vessel walls, and a thus a mutation at any of these sites results in the formation of cavernous malformations. In affected families, CCM1 is the most common mutation (40% to 50%), followed by CCM2 (30% to 40%) and CCM3 (about 5%). The CCM3 variant has the most aggressive clinical course.
It is important to emphasize, that patients with the spontaneous form of cavernous malformations have no apparent risk of passing the disease on to their children.
Diagnosing CCMs: The introduction of magnetic resonance (MR) imaging in 1986, revolutionized our understanding of cavernous malformations. Prior to the clinical availability of MR, the true incidence of these lesions was unknown. MR imaging is extremely good when it comes to demonstrating cavernous malformations. In addition, the lesions have a unique appearance on MR that allows most cases to be readily diagnosed without the need for surgical biopsy and pathologic evaluation.
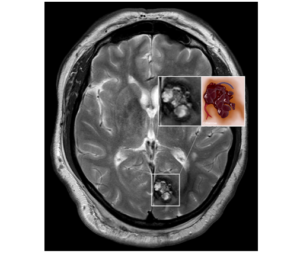
The classic appearance of cavernous malformations on MR includes a core that has a reticulated “salt-and-pepper” pattern and is surrounded by a halo of low (or absent) signal (Figure 2). Histologically, the core is composed of loculated areas of old-hemorrhage and thrombus (clot) of varying ages (Figure 2 inset). As was mentioned above, the walls of the blood vessels that form these lesions are abnormal and allow the breakdown products of the old-hemorrhages to readily escape into the surrounding brain. This is the same process that occurs when a bruise on your arm or leg is broken down leaving a temporary yellow stain in the skin that is gradually absorbed. In the brain this stain, which is caused by the breakdown of red blood cells, is not well absorbed. Instead the breakdown products gradually build up in the tissue surrounding the lesions and causes the loss of MR signal (the dark halo) that is so typical of their appearance (Figure 2 inset).
What is the natural history of CCMs? Natural history refers to the progress of a disease over time in the absence of any intervention. In order to make appropriate recommendations regarding management, physicians must understand the natural history of a disease. Only in this way can they decide if a potential treatment for the disease (no matter how effective it might be) involves more risk to the patient than the natural history of the untreated disease.
For CCMs the natural history is related to the location and presentation of the lesion. Cavernous malformations are found throughout the central nervous system (brain and spinal cord) in rough proportion to the volume of the various compartments with 80% located in the cerebral hemispheres, 15% in the deep brain structures and brain stem, and 5% in the spinal cord.
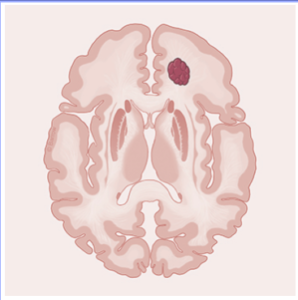
When the lesions are in the outer layers of the cerebral hemispheres (Figure 3), the most common presenting symptom is seizures. The seizures are thought to result from irritation to the brain caused by the buildup of breakdown products of red blood cells that sound the lesions, as discussed above.
When CCMs are present in deep brain structures and the brain stem, stroke like symptoms are the most common presentation (Figure 4). The brain stem gives rise to nearly all the nerves that supple motor and sensory function to the face, allowing us to move our eyes, hear, smile, frown, eat and speak. In addition, it also controls breathing. The brain stem and deep brain structures also serve as the pathways for all the motor and sensory fibers passing back and forth from the spinal cord to higher levels of the brain. So, in contrast to the cerebral hemispheres, symptoms from cavernous malformations in the brain stem and spinal cord typically produce changes in neurological function that mimic a stroke.
As was noted above, while CCMs are quite common in the general population, most of these lesions are silent. With the growing availability MR imaging, an increasing number of these asymptomatic (or incidental) CCM lesions are discovered when diagnostic studies are performed for other indications, such as the workup of headaches and head injuries.
What is the risk of symptomatic hemorrhage: Incidental CCM lesions have the lowest risk of causing hemorrhage. The risk of symptomatic hemorrhage for incidental lesions located in different areas of the brain are provided below in Table 1. For incidental lesions in the cerebral hemispheres the risk of symptomatic hemorrhage is no more than 0.1% to 0.5% per year; meaning that if we followed 1,000 affected patients for one year, we would expect the new onset of symptoms in 1 to 5 of those individuals.
Risk of Symptomatic Hemorrhage from Incidental CCMs
|
Location |
Risk of Symptomatic Hemorrhage |
|
Cerebral hemispheres |
0.1% to 0.5% per year |
|
Deep brain or brain stem |
2.5% to 6.8% per year (average 4.5%) |
|
Table 1: Risk of first symptomatic hemorrhage for incidental (asymptomatic) CCMs by lesion location |
|
While incidental lesions pose only a small risk to the individual, once a lesion has become symptomatic the risk of recurrent symptoms is increased about five times (5x). The risk of recurrent hemorrhage is again dependent on the location of the lesion. This is especially true of CCMs located in the brain stem where hemorrhages outside the lesion are common and the risk of recurrent hemorrhage is as high as 60% per year (see Table 2).
Risk of Recurrent Hemorrhage from Symptomatic CCMs
|
Location |
Prior symptom |
Risk of Recurrent Hemorrhage |
|
Cerebral hemispheres |
Seizures without hemorrhage |
1% to 4% per year |
|
Cerebral hemispheres |
Seizures with hemorrhage into surrounding brain |
25 to 30% per year |
|
Deep brain or brain stem |
Hemorrhage into surrounding brain |
30 to 60% per year |
|
Table 2: Risk of recurrent hemorrhage for previously symptomatic CCMs by lesion location |
||
Treatment options for patients with CCM: Presently, treatment options for the management of CCMs are limited to observation, observation with antiseizure medications, and surgery. Recommendations for treatment are based on lesion location and prior history. For those patients with incidental (asymptomatic) lesions the most appropriate management is observation regardless of location.
Observation is also considered reasonable for lesions in the cerebral hemispheres that present with seizures but are well-controlled with antiseizure medications. When seizures are poorly controlled, or the presentation is complicated by evidence of hemorrhage into the surrounding brain, surgery becomes an option. Surgery for these superficial, hemispheric lesions carries relatively low risks of complications in the range of 3% to 5%, and successful removal leads to the elimination, or improved control, of seizures in up to 70% of patients.
Observation is also the initial treatment of choice for incidental CCMs located in deep brain and brain stem locations. Even when patients present with evidence of hemorrhage outside the lesion, a trial of conservative management is usually recommended, unless the hemorrhage is associated with life-threatening clinical deficits. Likewise, conservative management is recommended for patients that have made a complete, or near complete, recovery from their prior bleeding episode(s). Surgery for deep brain and brain stem CCMs is typically reserved only for those patients that present with repeated episodes of symptomatic hemorrhage and progressive neurologic deficits. When considering surgery, it is important to remember that even in the best of hands, the operative removal of these brain stem lesions has major risks.
In a series of 300 consecutive cases operated on at the Barrow Neurological Institute, existing deficits worsened, or new deficits developed in 137 patients (53%) that underwent the surgical removal of deep brain and brain stem lesions. While many of these postoperative deficits improved, or resolved, with time, new deficits persisted in 93 patients (36%). Despite these risks, surgery in patients that have suffered multiple hemorrhages, or present with progressively more severe deficits appears warranted.
In properly selected patients, surgery may result in the more rapid recovery of deficits causes by a recent hemorrhage, and it also offers the potential of eliminating the risks of future bleeding episodes.
Future treatment options for CCMs: With the exception of anti-epileptic medication given for the management of seizures, there are currently no available medical treatments for the management of CCMs. A safe and effective systemic treatment that reduces or eliminates the risks of recurrent hemorrhage and leads to lesion dormancy is urgently needed.
Laboratory research has defined some of the key pathways involved in the formation of these lesions and resulting hemorrhage. Novel new agents are being pursued by the pharmaceutical industry that have the potential to block development and/or minimize the risk of recurrent bleeding. These drugs are still in various early stages of early development and will likely require 10 or more years to reach market.
The repurposing of several drugs that are currently in use for other clinical indications, offers potentially more rapid approval. These include the cholesterol lowering medication atorvastatin, and the blood pressure medication propranolol. Both drugs are being explored for proof of concept effect, and if effective could be available in as little as 2 to 3 years.
In future segments of this series we will look further at the evidence supporting the potential benefit of atorvastatin and propranolol in the treatment of CCMs, as well as some of the other exciting laboratory and clinical research being carried out to gain further insight into the development of cavernous malformations and the factors that control their natural history.
Summary:
- Cavernous malformations are much more common than generally appreciated, affecting about 1 in every 200 adults.
- While more than 12 million people in the United States are affected by cavernous malformations, only about 200,000 have suffered a symptomatic hemorrhage (bleeding episodes).
- Once a lesion has manifested a symptomatic hemorrhage, its untreated clinical course is quite serious, with a high risk of recurrent bleeding.
- Surgery in properly selected patients may speed the recovery from recent hemorrhage and offers the potential of eliminating risks of future bleeding episodes, but it not without serious risks.
- A safe and effective medical treatment that reduces, or eliminates, the risks of recurrent hemorrhage is urgently needed.

Comments 1
Kristen Freed
I just wanted to thank you for writing this article. I especially liked the sentence comparing a bleed to the yellow of a bruise. I met with my neurologist today and she told me I had a cavernoma and to be honest, the whole bleeding aspect of it freaked me out a bit as I tend to equate bleeding with hemorrhage as in game over. Framing it the way you did in the article, I gained a much better understanding.